Wenzhou TOONE Biotechnology Equipment Co., Ltd. has been deeply engaged in the field of microbial testing for more than ten years. As a National High-Tech Enterprise and one of China’s core suppliers of sterility testing consumables and equipment, TOONE is committed to the philosophy of “Building the Brand with Quality, Protecting Safety with Expertise.”
Focusing on microbial quality control needs in the pharmaceutical, food, CDC, and chemical industries, TOONE provides a complete range of solutions integrating sterility testing pumps, disposable sterility test canisters, and filtration consumables. All products comply with the standards of the Chinese Pharmacopoeia, deliver performance comparable to international brands, and offer outstanding cost-effectiveness.
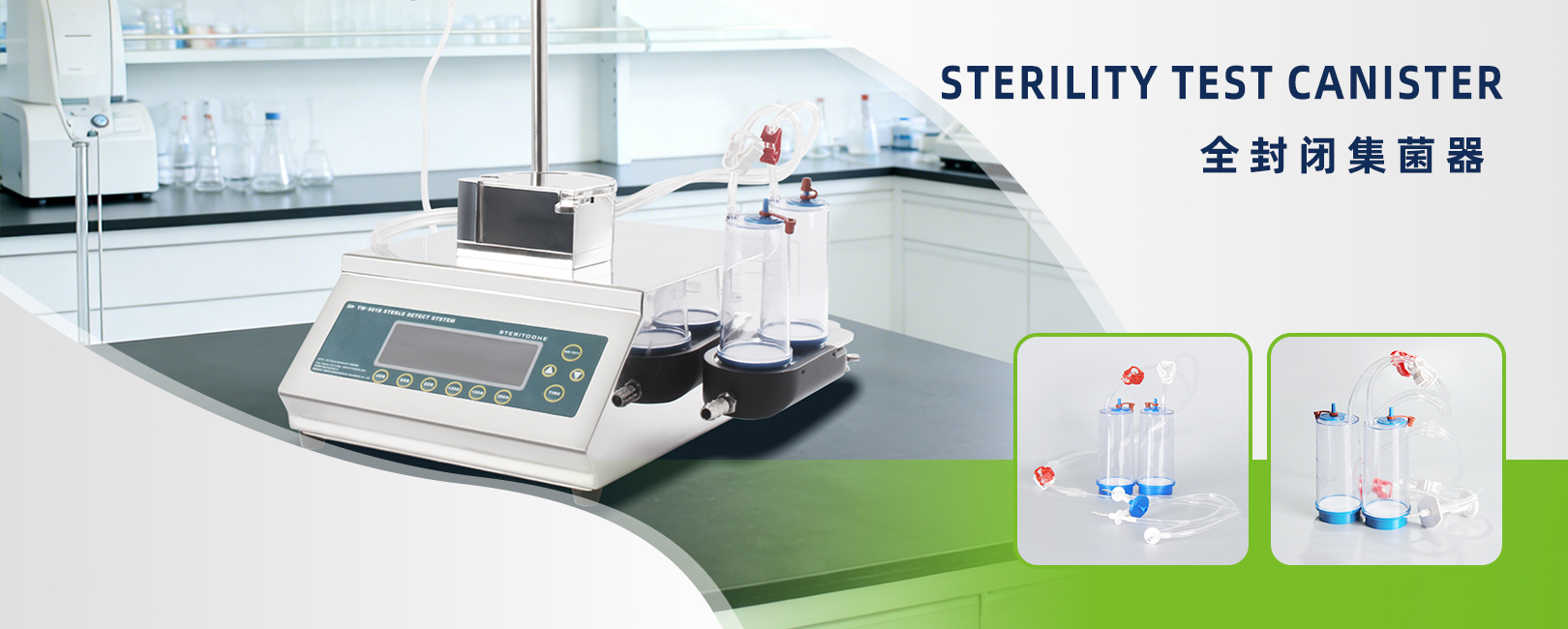
TOONE fully enclosed sterility test canisters (compatible with intelligent sterility testing pumps such as TW-APL01) are designed based on membrane filtration technology for sterility testing of pharmaceuticals, biological products, sterilized medical devices, and more. The system enables fully enclosed filtration with no cross-contamination and highly reliable results.
Key AdvantagesFully Enclosed Design:Integrated sterile tubing allows filtration, rinsing, media addition, and incubation to be completed within a completely closed system, effectively preventing external microbial contamination.
Safe and Durable Materials:The main body is made of AAS material, offering excellent cleanability and corrosion resistance.
Strong Compatibility:Compatible with single-channel, dual-channel, and multi-channel drainage systems, adapting to various sterility testing pumps and meeting both laboratory-scale and batch-testing requirements. Standard configuration includes a 0.45μm sterile membrane filter with ≥99.99% retention efficiency.
Convenient Operation:Combined with automatic pump control, culture media can be added directly after filtration for in-place incubation without transferring the membrane, significantly reducing contamination risk and improving counting accuracy and testing efficiency.
Applications
Pharmaceutical Industry
Sterility testing for injections, lyophilized powders, sterile APIs, and infusion products.
Medical Devices
Sterility validation for disposable syringes, infusion sets, and surgical instruments.
Biological Products
Sterility quality control for vaccines, sera, and culture media.
II. Star Consumables for Microbial Limit Testing: TW-M50 / TW-F100 / TW-M50B — Efficient, Accurate, and Reliable
1. TW-M50 Disposable Microbial Limit Test Unit — The Convenient Choice
Ready-to-Use
Pre-installed sterile membrane filter, EO sterilized (SAL ≤10⁻⁶). No cleaning or sterilization required before use.
High Filtration Efficiency
Uniform high-porosity membrane provides fast flow rates and resists clogging, making it suitable for high-viscosity samples while ensuring complete microbial retention.
Applications
Microbial limit testing and quality control for:
Food & beverages: purified water, mineral water, beverages, dairy products
Pharmaceuticals: oral preparations, capsules, tablets, APIs
Cosmetics: creams, lotions, personal care products
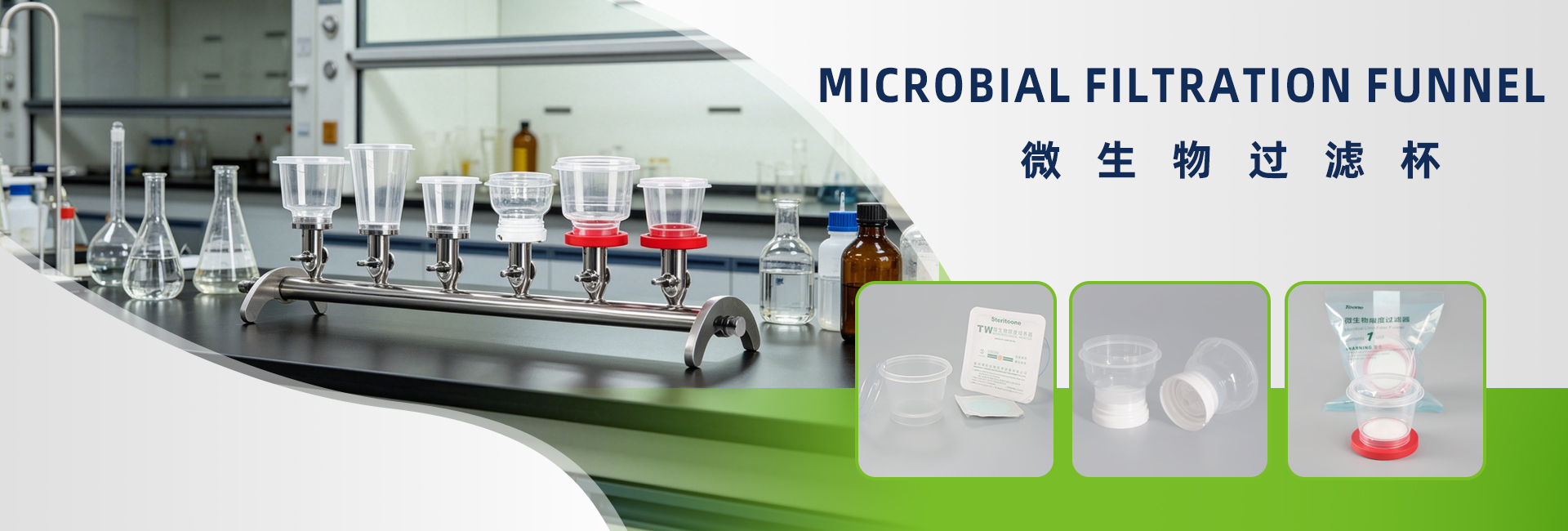
2. TW-F100 Reusable Microbial Filtration Funnel — Durable and Cost-Effective
Long Service Life
Made from premium materials resistant to repeated high-temperature sterilization, aging, and corrosion.
Flexible Compatibility
Compatible with multiple filtration manifolds and microbial limit testing systems; supports different membrane pore sizes for diverse testing requirements.
Lower Operating Cost
Significantly lower per-test cost compared with disposable products, ideal for high-frequency and large-volume laboratory testing.
3. TW-M50B Disposable Sterile Filter Cup — Closed Filtration with Accurate Counting
Simple Operation
Disposable base design eliminates repeated flame sterilization, greatly improving workflow efficiency.
Applications
Pharmaceutical companies: purified water, WFI, oral preparations
Food manufacturers: semi-finished products, finished goods, packaging materials
Third-party laboratories: high-volume microbial limit and contamination testing
III. Four Core Reasons to Choose TOONE Microbial Testing Consumables

1. Regulatory Compliance and Reliable Quality
Products comply with the Chinese Pharmacopoeia, GMP requirements, and food hygiene standards. Strict sterility validation and quality testing ensure dependable performance, with multiple technical parameters incorporated into industry standards.
2. Advanced Technology and Outstanding Performance
Fast membrane flow rates, excellent microbial retention, fully enclosed anti-contamination designs, and safe non-toxic materials suitable for high-temperature sterilization.
3. Complete Product Ecosystem
A comprehensive product line including sterility test canisters, microbial limit test units, filter cups, manifolds, and sterility testing pumps provides one-stop solutions for sterility testing, microbial limit testing, and membrane filtration applications.
4. Excellent Cost Performance and Professional Service
As a high-quality domestic brand, TOONE offers exceptional value along with nationwide technical support and after-sales service to ensure worry-free laboratory operations.
From the stringent quality control of sterile pharmaceuticals to the safety assurance of food and beverages; from CDC environmental monitoring to precise laboratory research, TOONE microbial testing consumables consistently deliver professional quality, innovative design, and reliable performance for every test.
Choosing TOONE means choosing a precise, safe, efficient, and economical microbial testing solution that helps your products and laboratories successfully pass every quality challenge.